Fusion-relevant materials
The progress in the development of fusion power plants is closely linked to the availability of suitable materials for structural parts that are exposed to extreme conditions. For the most thermally loaded part, the divertor, in which the proposed operational temperature is in the range of approx. 800-1400 °C, tungsten-based composites were selected as candidate materials.
To overcome the main drawbacks of the currently available composites, within the European fusion programme EUROfusion we explore two options for improvement:
- dispersion strengthening of W with small W2C particles formed during the sintering by reaction of W with the precursor and
- development of new WC-based cemented carbide containing low-activation high-entropy alloy as a binder phase.
Contact:
Solar energy materials
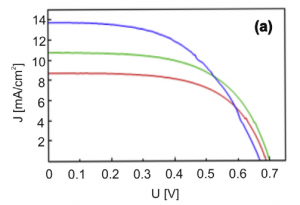
One-dimenzional TiO2 nanotubes were used as photoactive materials in classical rigid and flexible dye-sensitized solar cells (DSSC). Self-ordered, vertically aligned, titanium oxide (TiO2) nanotubes were grown on an electropolished titanium foil by anodic oxidation in a viscous organic electrolyte. Different anodization times were used in order to obtain nanotubes with length between 10 and 17 µm. To improve the sensitizing of the high-aspect-ratio nanotubes with dye we used a new approach in which the TiO2 arrays were immersed into the dye solution at elevated temperature in an autoclave. The current–voltage characteristics of the assembled DSSCs gave an average energy-conversion efficiency of 5.7% for the untreated nanotubes.


Another approach to obtain photoactive material with enhanced properties was by hydrothermal synthesis of self-assembly of rutile mesocrystals which resulted in formation of hierarchical nanostructures. Superior efficiency in photo-electrochemical production of hydrogen and improved solar energy conversion efficiency was demonstrated.
Contact:
N-type oxide thermoelectric ceramics
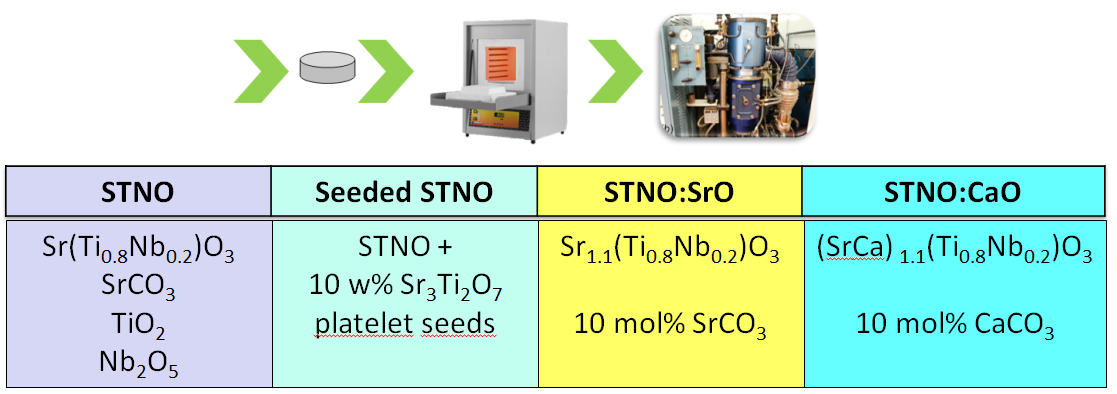
Figure of merit (ZT) of oxide-based TE materials can be improved by introducing planar faults into the microstructure. It is expected that in-grown planar faults reduce thermal conductivity of the material which consequently increases the ZT value of the material. This approach to reduce thermal conductivity was used in n-type Sr(Ti0.8Nb0.2)O3 (STN) thermoelectric materials to which Ruddlesden-Popper (RP) type faults were added via Sr3Ti2O7 platelet crystals or SrO and CaO. The observed RP planar faults in STN matrix possess rock salt-type structure and form either random 3D networks along {001} STN crystallographic planes and/or more or less ordered polytypoidic sequences with general formula (Sr,Ca)O*n(Sr(Ti,Nb)O3). The thermoelectric measurements showed that thermal conductivities of doped STN samples were app. twice lower as compared to undoped STN. Consequently, the figure of merit (ZT) for e.g. CaO-doped STN was improved by a factor of two.


Contact:
Ca3Co4O9-based p-type thermoelectric ceramics
The Ca3Co4O9 compound (Ca349) is p-type thermoelectric material with complex misfit layered structure, distinct plate-like morphology of grains and consequently largely anisotropic physical properties (i.e. in single crystal electrical conductivity is about 5-times larger in a-b planes than along the c-direction).
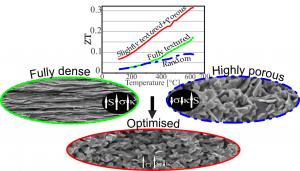
Accordingly, microstructure can have has strong influence on the thermoelectric characteristics. The influence of processing method (classical sintering, hot pressing, SPS, cold pressing) on microstructure of the Ca349 ceramics, morphology of grains, density and degree of texturing, and consequently their TE characteristics was studied.
In contrary to general considerations in literature that fully dense and perfectly textured microstructure would result in optimal thermoelectric characteristics, we obtained the highest ZT of 0.31 at 600°C reported so far for the pristine Ca349 compound in modestly textured and high porosity ceramics; at several times lower thermal conductivity it had similar Seebeck coefficient and electrical conductivity as fully dense and perfectly textured Ca349 ceramics.
Contact:
Thick-film thermoelectric microgenerators
An alternative to the expensive and time-consuming processing of the bulk thermoelectric microgenerators (TEG) for waste heat harvesting by direct conversion into electricity and sensor applications could be the structuring of TEG by means of screen-printing technology. Two types of thermoelectric microgenerators (TEGs) made by screen printing on alumina substrate, one from a Ca3Co4O9 (Ca349) p-type leg and the other from a (ZnO)5In2O3 (Z5I) n-type leg, are developed. Our study defines a promising fabrication method for the oxide-based thermoelectric devices and also identifies the main challenges associated with the microgenerator fabrication.

Contact:
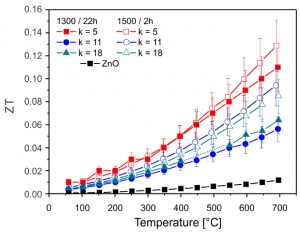
ZnO-based n-type thermoelectric ceramics
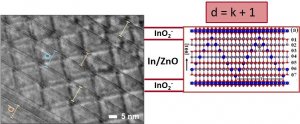
The ZnO-based ceramics is promising n-type oxide thermoelectric for high temperature applications. ZnO ceramics has very high Seebeck coefficient (S) of about -400mV/K; however it also has low electrical conductivity (s) of about 5S/cm and much too high thermal conductivity (k) of about 40W/mK for good thermoelectric characteristics, which are expressed by dimensionless figure of merit ZT (ZT = sS2T/k). Hence, focus of our research is on development of the ZnO ceramics doped with various dopants, which can significantly enhance their electrical conductivity and reduces thermal conductivity, such as oxides of In, Ga and Al, and also others (Fe, Y, Nd, Ce and Nb). A complex defects can be introduced into relatively simple wurtzite-type structure of ZnO by doping with In2O3 and/or Ga2O3, and their ordering result into ZnkM2Ok+3-type homologous phases. While (MO2)–-rich layers with free charge carriers provide highly conductive channels, zig-zag M/ZnO defects in between planar defects effectively scatter phonons and reduce thermal conductivity. Hence, the ZnkM2Ok+3-type homologous phases represent interesting platform for tailoring thermoelectric characteristics of the n-type ZnO-based ceramics.
Contact:
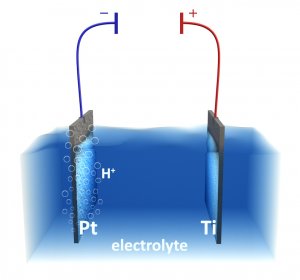
Photo(electro)catalytic reactors
The group has designed and constructed new original photo(electro)catalytic reactors based on fixed photoactive material for wastewater treatment (removal of organic compounds), gas treatment (removal of VOC) and/or selective synthesis of organic molecules. So far many organic test compounds have been successfully completely mineralized. Scale-up of the 3rd generation reactors are currently in progress.


Luka Suhadolnik et al, Journal of industrial and engineering chemistry, 2017, vol. 47, 384-390
Luka Suhadolnik et al, The chemical engineering journal, 2016, vol. 303, 292-301
Luka Suhadolnik et al, Solar energy, 2016, vol. 127, 232-238